Currently, for patients suffering from lower back pain in the US and in Europe cell therapies that are available employ the “classic” stem cells. These are stem cells that are isolated for example from the bone marrow or fat tissue. They are being broadly applied in several fields of medicine with variable success. These cell therapies are intended to alleviate pain by harnessing inflammatory processes ongoing in the diseased disc tissue. However, these cells do not restore the tissue and as such are expected to only have temporary effects. iPSpine focuses on generating a therapy where the advanced stem cells harness inflammation and pain. In addition, the iPSpine stem cells will have the ability to rejuvenate the disc tissue. The advanced stem cells are supported by the smart biomaterials that provide them a safe home to thrive within the harsh disc environment.
We target the iPSpine therapy to be a one off injection with long lasting effects for more than 1 year. Pain alleviation should be evident already on the short term (first weeks upon injection), while the rejuvenating effects should become visible by MRI at 1 year follow up and even later. The delayed effect of rejuvenation relates to the fact that the injected advanced stem cells need to thrive, multiply and produce new tissue. This is a process that occurs slowly within the disc due to limited nutrition available to the disc cells as compared to, for example, the skin where blood supply is ample and healing occurs within a few weeks at most.
Once developed, the iPSpine treatment is meant to be a single-day treatment. The patient will receive a local anesthetic to numb the procedural area. Using X-ray guidance the specialist will determine the best and safest way to access the diseased disc using a thin needle. The needle is inserted under X-ray guidance into the center of the disc. Once the specialist is sure that the needle is on target, he/she will inject a small volume of the biomaterial containing the advanced stem cells. Thereafter the patient can recover from the procedure in about 30-45 minutes and is allowed to go home.
Indeed the iPSpine therapy is intended to reduce (or delay) the chances of recurrence of lower back pain and to prevent the need for surgical treatment.
Your own stem cells will not be used for the iPSpine treatment. Using your own stem cells would imply that these need to be reprogrammed into iPS cells, thereafter differentiated into the juvenile disc cells. All these steps would have to be extensively characterized and checked for quality as an advanced medicinal product. As such, personalized iPS cell therapies for diseases affecting large population of patients, like lower back pain patients, will be too expensive, have limited availability and most possibly, as such, not reimbursed by the health care systems.
The iPSpine treatment has an initial starting point to be a treatment for patients with a degenerated disc. An intact disc, though degenerated, should initially be considered as a prerequisite for the treatment to stay in place after injection and such be safe for the patient. If the disc is not intact, there is a risk for the hydrogel to dislocate out of the disc and a possible risk for an immune reaction to the “foreign” iPS juvenile disc cells. Also, an intact disc also ensures that there are no immune cells already present in the disc that can cause an immune response.
Herniated discs are treated conservatively (e.g., pain medication, physiotherapy) and, depending on the health care guidelines of each country, surgically removed in situations that the herniated disc causes severe pain and harm to the nerve tissue being compressed resulting into neurologic deficits. On the long term, in both treatments (conservative and surgical) there will be repair tissue formed to close the gap in the outer layer of the herniated disc. It is expected that these repaired degenerate discs could also be treated with the iPSpine therapy. However, it remains to be determined if there are other risks (e.g., re-herniation on the longer term).
Patients suffering from certain nerve root compression causes may not benefit as optimal from the iPSpine treatment. Both herniated disc and spondylolisthesis (“slipped disc”) would be difficult to treat due to the chances in local geometry of the disc. The iPSpine therapy is injected locally in the center of the disc. From a biological perspective, there is a crosstalk between tissues locally, and if there is inflammation in a tissue, other tissues can sense this and generate pain. The iPSpine therapy is at its best when used at the earlier stages of disc degeneration, i.e., before considerable facet joint degeneration and/or spondylolisthesis occurs.
Once proof-of-concept has been established for treating degenerated discs, more in-depth studies can be conducted to identify other patient populations that may benefit from the treatment.
Yes, the advanced stem cells being developed are derived from human. The advanced stem cells in this project are called induced pluripotent stem cells. This is abbreviated as iPS cells. iPS cells are generated in the laboratory.
White blood cells are programmed in the laboratory to revert from a fully developed state back to embryonic stem cells. iPS cells can multiply themselves well, making them ideal to make enough cells for the large population of patients suffering from degenerative discopathy. The iPS cells are re-programmed to notochordal cells, the potent cells only present in young and healthy discs.
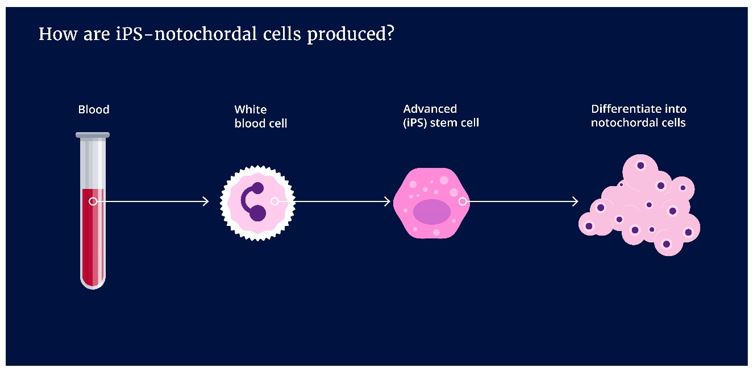
It is not forbidden by law. We do not use human notochordal cells because these would need to be collected from human embryos and children. Next to the ethical concerns, such sources are not broadly available for the millions of patients that need treatement against back pain. Furthermore, to date, the available technology does not support amplification of human notochordal cells in the laboratory. The notochordal cells loose their identity when being amplified.
In patient care, preventive treatments are only considered as a possibility once they have proven efficacy to work. The iPSpine approach that is being developed is intended for therapeutic purposes. The project encompasses laboratory and preclinical work to further develop the envisioned advanced therapy. The project focuses on addressing biology questions that help us understand the mode of action of the advanced stem cells and their interaction with the biomaterials and the disc tissue where they will be implanted. The foremost activities are centered on safety and possible efficacy of the engineered cells in combination with the smart biomaterials.
There are additional aspects that would need to be addressed to consider the developed strategy for preventive therapy in patients that may be at risk to develop disc degeneration. The concerns about safety there are even more stringent and carry ethical challenges.
A difficulty with understanding whether a treatment could be used preventively is that it needs to be tested for multiple years in order to get insights in uncertain and unknown side-effects and whether it is effective as a preventive treatment. Only once a wide population of patients has been treated may these scarce and expected side effects be observed. The difficulty lies in how severe a person’s lower back pain will become if he or she will not be treated and what the long-term differences are between having a preventive iPSpine treatment, an alternative treatment and no treatment. Such research will as well bring insights in whether iPSpine as a preventive treatment is proportional in terms of efficacy and side-effects for the (future) pain it is trying to relieve or alleviate. Another consideration would be what the alternatives are. When there are alternatives with fewer side-effects, that are less invasive and/or financially less costly, it could be possible that these alternatives will be preferred. The availability of iPSpine as a preventive treatment will as well depend on whether health insurances will cover the costs of a preventive treatment when other healthcare costs might be more urgent. Altogether, these considerations imply that the iPSpine treatment needs initially be considered as a therapeutic application once clinical signs are at play.
NC Biomatrix is a company that is commercializing the use of a molecular treatment product for disc degeneration that is based on the tissue found in the center of pig discs. iPSpine is exploring the use of a similar base material in combination with other biomaterials and with advanced stem cells which in essence are notochordal cells engineered in the laboratory with technology that the iPSpine project develops. iPSpine hopes that the base material will help to recapitulate the healthy environment and provide the engineered cells with safe cues which flourish within the disc. Also by combining it with other biomaterials we hope to enhance its biomechanics restoring properties. The product of NC Biomatrix is a simpler approach which can be translated more quickly but will only address individuals with early to moderate disc degeneration. It may also be used prophylactically. The iPSpine product is more complex and will take much longer to reach the clinic but is inherently more powerful and will address a much larger population of patients with even severe disease because of its use of cells.
If a disc is herniated, there is a chance of the hydrogel to leak out. Technically the immune system will be exposed to a foreign body material, i.e., the “foreign” iPS juvenile disc cells, and a local immune reaction may occur. Therefore, the treatment is not suitable for patients suffering from an acute herniated disc.
The iPSpine project starts with the understanding that as long the disc is intact the disc environment remains protected from the immune system. The degenerate disc is considered intact if the content from the center of the disc has not herniated through the outer layer. As such, it is expected that the immune system will not get in touch with the “foreign” iPS juvenile disc cells. While iPSpine will study these iPS juvenile disc cells in clinically relevant animal models, parallel research activities focus on whether it is possible to bypass a possible immune response. The first steps are undertaken in the laboratory to also study modified iPS cells that have been engineered to not elicit an allergic response even if they would come in contact with the immune system.
No, it is not possible yet to subscribe for a Clinical Trial of this project. Currently, for patients suffering from lower back pain in the US and in Europe stem cell therapies that are available employ the “classic” stem cells. These are stem cells that are isolated for example from the bone marrow or fat tissue and are being broadly applied in several fields of medicine with variable success. In Europe, the RESPINE project, subsidized by the EU, is recruiting and treating the first patients. We collaborate with partners from RESPINE in order to make a swifter step towards the clinic when the advanced iPSpine therapy is ready.
This will take at least five project years (2024). iPSpine aims to develop an advanced therapy combining the specialized stem cells and biomaterials to enable the cells to thrive in the harsh disc environment. These type of therapies require long development trajectory demonstrating their safety and possible efficacy before they can be transferred to the patient. The therapy being developed in the iPSpine project is in the beginning stages of such a trajectory. Extensive laboratory research will be followed by research with laboratory animals to demonstrate that this advanced therapy is safe. In five years we will know if the treatment is promising. If so, we will then start preparing for the first study in human patients.
You can subscribe for the patient feedback group. We are now looking for international, motivated individuals with chronic lower back pain to join our iPSpine patient feedback group to support the Patient Advisory Board (PAB) activities. We are looking for individuals who: have experience living with, or have been diagnosed with chronic lower back pain, and who want to help us gain the patient perspective. The feedback group will support our Patient Advisory Board (PAB) consisting of patients, a clinician, an ethicist, and patient engagement researchers.
As part of our feedback group, you may be asked to participate in community outreach (e.g. social media posts, spreading the message about the project) and/or review study materials made available to the public (e.g. website content, lay summaries of publications, etc.). If you are interested to join, please send an email to ipspine@catalyze-group.com.
The more patients involved in the project, the more impactful the project will be!
The iPSpine treatment is currently under development and not yet available for patients. Consult your treating physician for questions regarding your personal treatment options.
We are in an early stage of research and the answer to the specific questions below is unclear. We will know more around 2021-2022 and will be able to address these questions more adequately.
- How soon will the treatment be available?
- Is there a financial cost to the patient?
- Will it improve my quality of life?
- Is it safe? Are there any side effects?
- Can you have repeated injections if the condition is degenerative?
- Will it be embraced by the NHS?
- Where would I need to go for treatment?
- Are there any contra indications that would make an individual unsuitable for treatment?
- How will patients be identified as suitable for this treatment?
- How long will the treatment last, is it a one off injection or a course of treatment?
In all vertebrates there is structure resembling a flexible stock that extends throughout the body of the embryo. This structure is called the notochord. The notochord ensures firmness and gives signals that drive the development of the embryo. It is temporary; the notochord disappears and at some point, when the development of the spine is complete, notochordal cells only reside is in the center of the disc.
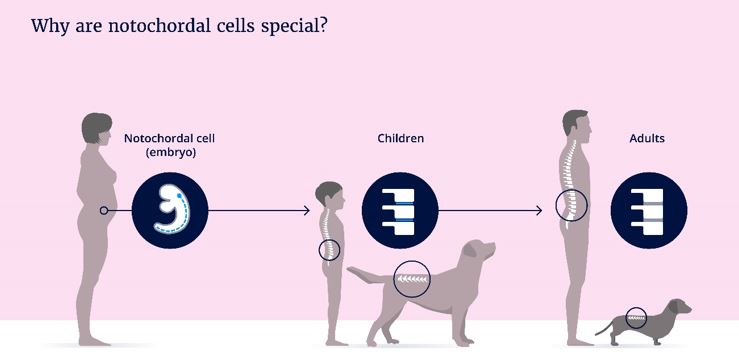
There is something very special about notochordal cells.
Notochordal cells are present in children but disappear later in life. It happens, to what extent it is a coincidence, that 80% of people suffer from back pain once in their lives . Similar problems occur also in our 4-legged family-members; the dog. The problem mostly occurs in dog breeds with relatively short legs, such as the Dachshund, that often suffers from a acute hernia early in life. In other dog breeds, such as the Labrador, the notochordal cells remain present in adult animals and we only see back problems later in life.

